Anti-CD40 Colitis in Mice

Induction of Anti-CD40 Colitis in Mice:
On study day 0, female C57BL/6 Rag2-/-(n12) or CB-17 SCID mice are injected (IP) with anti-CD40 IgGa monoclonal antibodies (mAb) to induce innate ulcerative colitis.
Disease Parameters:
Injection of an agonistic CD40 monoclonal antibody (mAb) to T and B cell-deficient mice has been shown to be sufficient to induce a pathogenic systemic and intestinal innate inflammatory response with intestinal inflammation associated with interleukin-23 (IL-23)(p19) mRNA-producing intestinal dendritic cells and IL-17A mRNA within the intestine1.
Dosing Paradigms:
- Begin dosing on study day 0 and continue until necropsy on day 7.
- Route of administration: SC, PO, IP, IV, IC
Clinical Assessment:
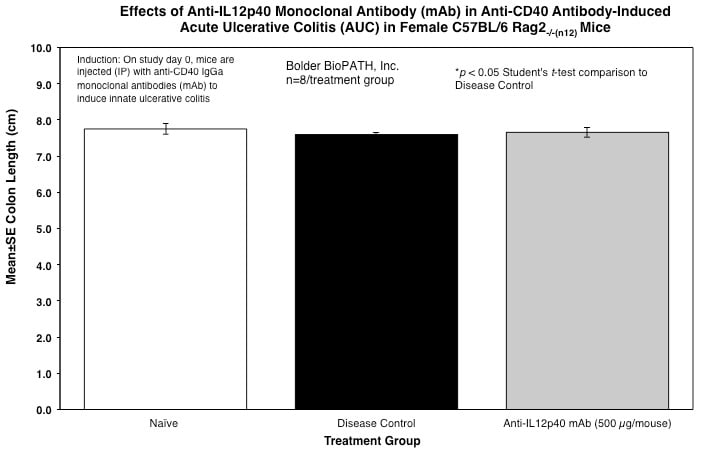
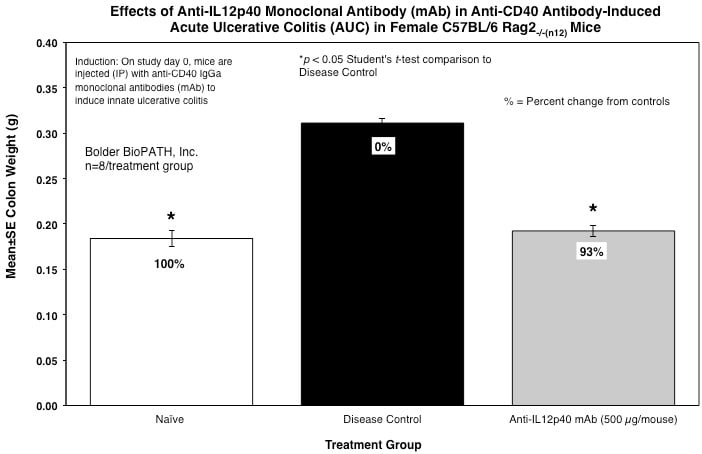
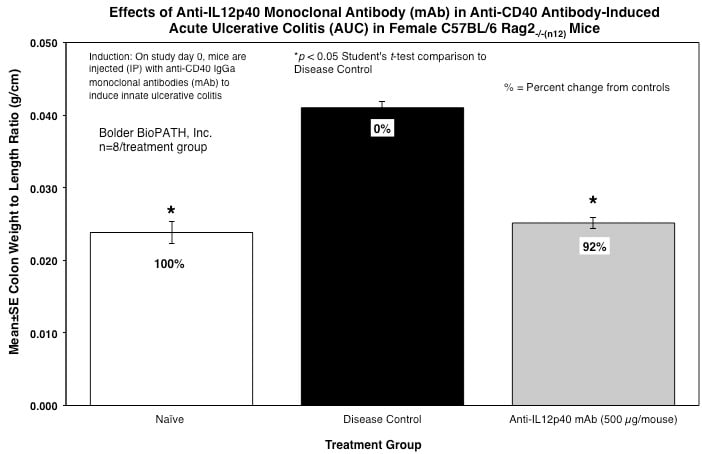
From each animal, the entire colon is harvested, inspected visually, measured for length, and weighed.
Histopathological Assessment:
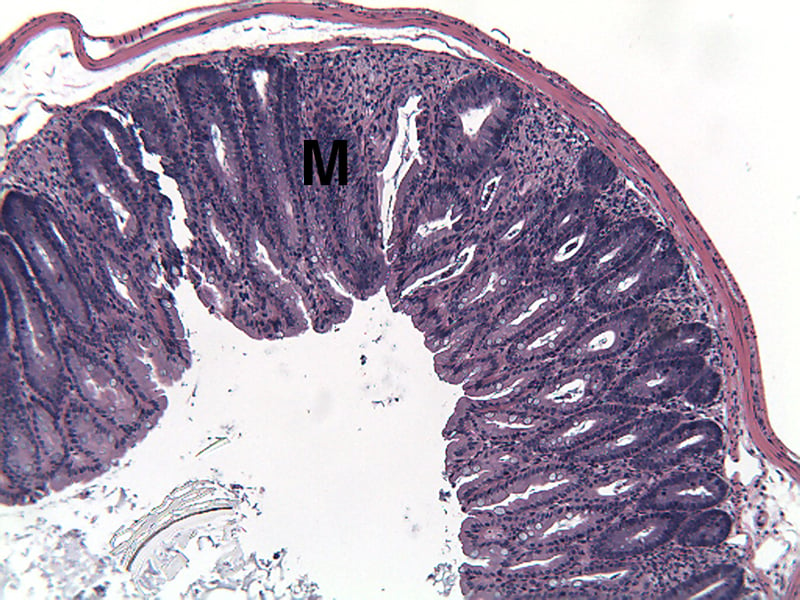
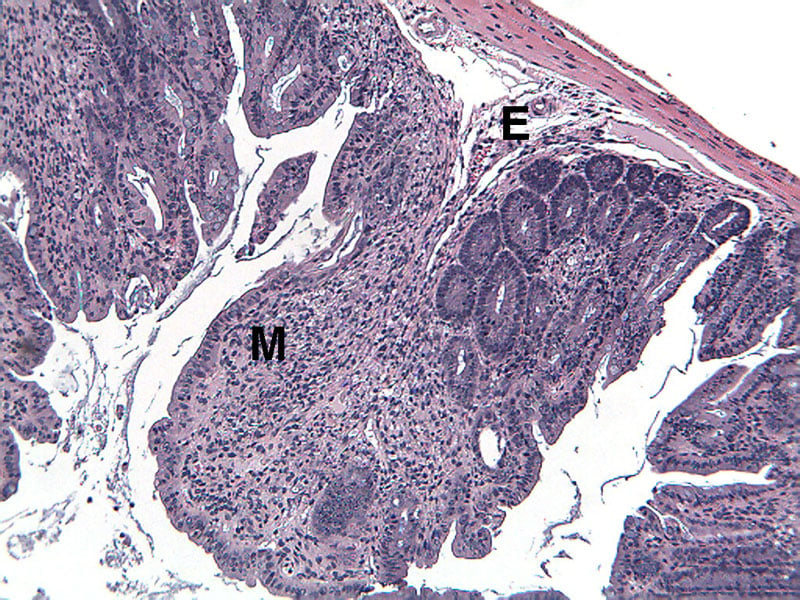
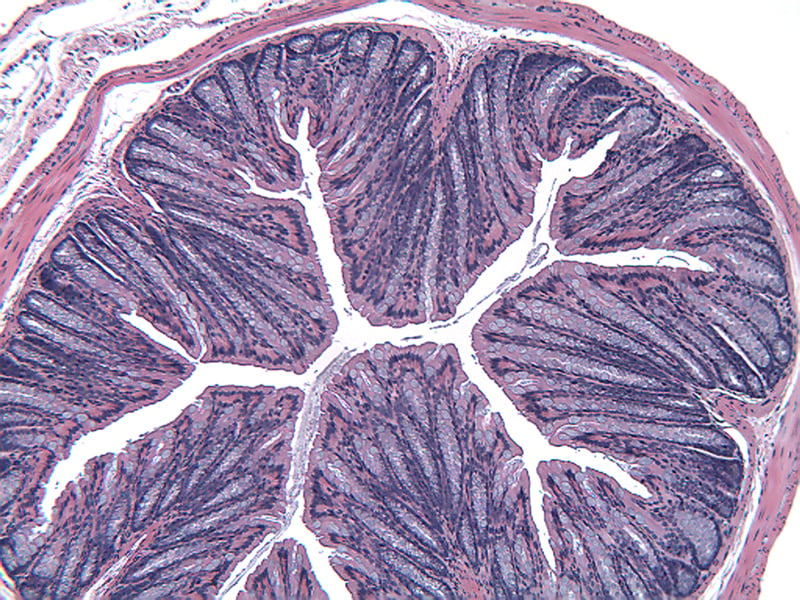
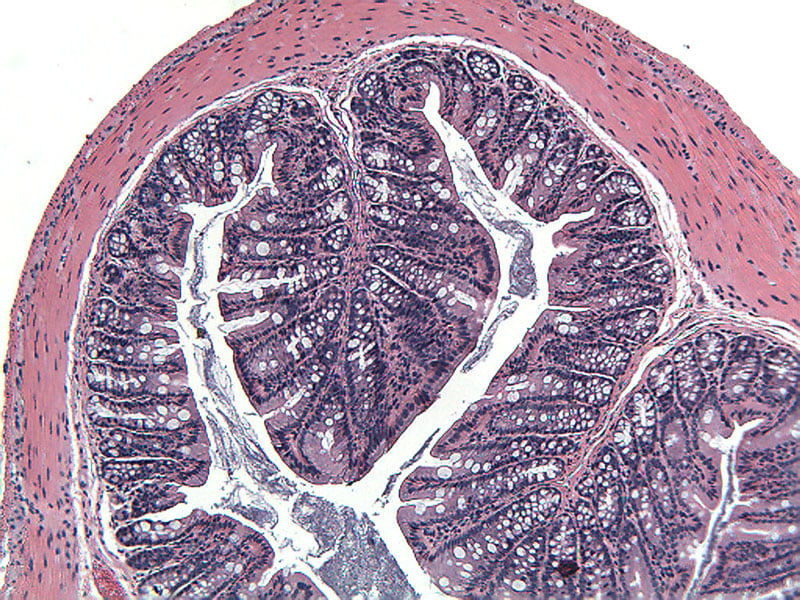
The colon is cut into Proximal and Distal halves and collected for processing and embedding. Each half is sectioned into 3 equidistant pieces and these sections are stained to quantitate inflammation, gland loss and epithelial loss (hematoxylin & eosin), which are scored according to these methods.
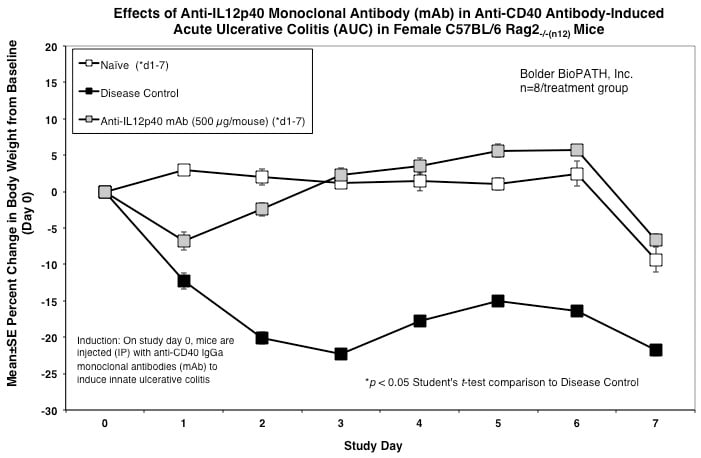
Sample Data (Click on image to enlarge):
Representative Photomicrographs of Colons
For additional examples of positive controls, please contact us.
Notes:
The CD40-CD154 pathway is important in the pathogenesis of inflammatory bowel disease (IBD), a gastrointestinal disorder that can manifest as Crohn’s disease (CD) or ulcerative colitis (UC). Injection of an agonistic CD40 monoclonal antibody (mAb) to T and B cell-deficient mice has been shown to be sufficient to induce a pathogenic systemic and intestinal innate inflammatory response with intestinal inflammation associated with interleukin-23 (IL-23)(p19) mRNA-producing intestinal dendritic cells and IL-17A mRNA within the intestine1. IL-23(p19)- or IL-23R-deficient mice are resistant to IBD, and the inhibition of IL-23 by anti-p19 neutralizing mAbs blocks organ-specific autoimmune inflammation. IL-23(p19), IL-12(p40), and IL-17A expression is elevated in human inflamed gut from CD and ulcerative colitis patients, and experimental and clinical data have pointed to IL-23 as a therapeutic target for the treatment of IBD2.
Optional Endpoint:
- Blood collection
- Feces collection
- Disease Activity Index (DAI)
- PK/PD blood collections
- Cytokine/chemokine analysis via Luminex(R)
- Other sandwich ELISAs
- CBC/clinical chemistry analysis
- Soft tissue collection
- Histopathologic analysis
- Immunohistochemistry analysis
- Endoscopy
References:
- Uhlig HH, McKenzie BS, Hue S, et al. Differential activity of IL-12 and IL-23 in mucosal and systemic innate immune pathology. Immunity. 2006;25:309–318. doi: 10.1016/j.immuni.2006.05.017
- Cayatte C, Joyce-Shaikh B, Vega F, et al. Biomarkers of therapeutic response in the IL-23 pathway in inflammatory bowel disease. Clin Transl Gastroenterol. 2012; 3(e10). doi: 10.1038/ctg.2012.2